Background
MAVORIC was a phase 3, open-label, multi-center, randomized controlled trial that evaluated the safety and efficacy of mogamulizumab compared with vorinostat in patients with relapsed/refractory mycosis fungoides (MF) or Sézary syndrome (SS) (NCT01728805). Mogamulizumab-associated rash was the second most common TEAE of any cause or grade in the mogamulizumab treatment arm and the most common TEAE leading to treatment discontinuation, resulting in a discontinuation rate of 7% (13/184). Grade 1-2 and 3 drug rashes occurred in 20% (36/184) and 4% (8/184) of mogamulizumab-treated patients, respectively. The objectives of this analysis were to describe histopathological and other characteristics of drug rash in patients who received mogamulizumab in MAVORIC.
Methods
In MAVORIC, 372 patients were randomized 1:1 to receive either intravenous mogamulizumab at 1.0 mg/kg once weekly for Cycle 1 (28 days) and then on days 1 and 15 of subsequent cycles or oral vorinostat at 400 mg daily. Confirmed overall response rate (ORR; complete response + partial response) was based on a global composite response involving all four disease compartments and verified at two consecutive visits. Guidance to investigators was provided in the protocol for evaluation and management of new drug rash on mogamulizumab treatment. Patients with an initial Grade 1 drug rash were allowed to continue treatment with use of topical steroids as needed. For patients with Grade ≥2 drug rash, mogamulizumab was temporarily stopped, and treatment of the drug rash with topical steroids was advised. Use of systemic steroids was prohibited during the study. Treatment could resume if the drug rash resolved to Grade ≤1 within 2 weeks. Biopsies were evaluated by an on-site pathologist, and a central blinded review was also conducted.
Results
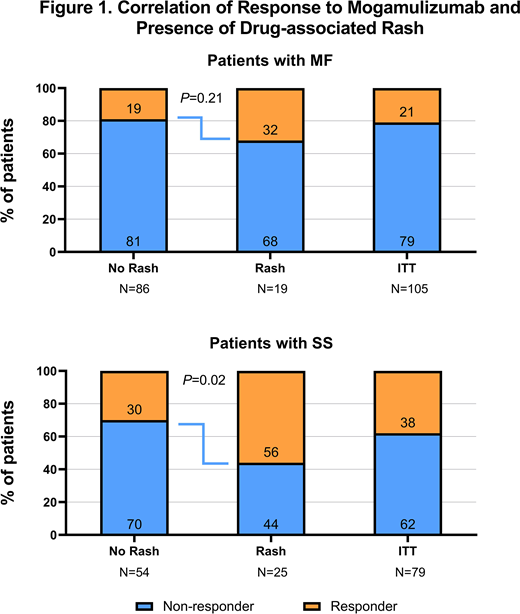
This analysis included 44 patients who were treated with mogamulizumab and experienced drug rash during MAVORIC. Pathologically, central review assessed rashes as granulomatous, histiocytic spongiotic, lichenoid, eosinophilic, psoriasiform, or a combination of these patterns with no clear predominant histological pattern. Among the 44 patients with drug rash, 59.1% (26/44) were ≥65 years of age, and more patients with SS [56.8 (25/44)] than MF [43.2% (19/44]) experienced drug rash. Median (Q1, Q3) exposure among patients with drug rash was 344 days (162, 652) in patients with SS and 185 days (85, 463) in patients with MF. Mogamulizumab did not result in any cases of anaphylaxis or SJS/toxic epidermal necrolysis (TEN). Concomitant or immediate prior CTCL therapies that may make patients prone to drug rash were examined, and no trend toward increased incidence of drug rash was observed with any particular agent or class. The proportion of SS responders with drug rash was significantly higher than responders without rash (P=0.02; Figure 1); the proportion of MF responders with drug rash did not differ from responders without rash (P=0.21). Overall, the median (Q1, Q3) time to onset of drug rash was 106 days (36, 254). Initial drug rash occurred after response to mogamulizumab in 70% (14/20) of patients who experienced both. Thirty-five patients (80%) in the drug rash cohort resumed treatment with mogamulizumab upon resolution of initial drug rash per-protocol. After these patients resumed treatment, the median (Q1, Q3) duration of exposure to mogamulizumab was 183 days (58, 332).
Conclusions
Mogamulizumab-associated rashes displayed heterogeneous histopathology without one predominant feature; therefore, close correlation of suspected drug rash with clinical features is advised to differentiate it from disease progression. A trend toward higher rate of drug rash was observed in patients with SS, the group more likely to respond to mogamulizumab. Therefore, drug rash may be attributable to increased exposure, but rash may also involve underlying disease characteristics or treatment-induced immune alterations; further investigations are warranted. Nevertheless, the observation that 80% of patients on trial were able to continue mogamulizumab for >6 months following resolution of their initial rash suggests that appropriate evaluation, identification, and management of these reactions may prevent premature discontinuation.
Musiek:Kyowa Kirin: Honoraria; Helsinn: Honoraria; miRagen: Other: Investigator; Elorac: Other: Investigator; Soligenix: Other: Investigator. Horwitz:ASTEX: Consultancy; Affirmed: Consultancy; Vividion Therapeutics: Consultancy; Verastem: Consultancy, Research Funding; Infinity/Verastem: Research Funding; Kyowa Hakka Kirin: Consultancy, Research Funding; Millenium/Takeda: Consultancy, Research Funding; Seattle Genetics: Consultancy, Research Funding; Trillium: Consultancy, Research Funding; Corvus: Consultancy; Innate Pharma: Consultancy; Mundipharma: Consultancy; Portola: Consultancy, Research Funding; Beigene: Consultancy; C4 Therapeutics: Consultancy; Daiichi Sankyo: Research Funding; GlaxoSmithKline: Consultancy; Janssen: Consultancy; ADCT Therapeutics: Consultancy, Research Funding; Forty Seven: Consultancy, Research Funding; Kura Oncology: Consultancy; Miragen: Consultancy; Myeloid Therapeutics: Consultancy; Aileron: Consultancy, Research Funding; Celgene: Consultancy, Research Funding. Bagot:Helsinn/Recordati: Membership on an entity's Board of Directors or advisory committees; Takeda: Membership on an entity's Board of Directors or advisory committees; Kyowa Kirin: Membership on an entity's Board of Directors or advisory committees; Innate Pharma: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding. Huen:Kyowa Kirin: Membership on an entity's Board of Directors or advisory committees, Research Funding; Medivir: Membership on an entity's Board of Directors or advisory committees, Research Funding; GlaxoSmithKline: Research Funding; Seattle Genetics: Membership on an entity's Board of Directors or advisory committees, Research Funding; Galderma: Other: Travel expense reimbursement, Research Funding; miRagen: Membership on an entity's Board of Directors or advisory committees, Research Funding; Helsinn: Membership on an entity's Board of Directors or advisory committees; Innate Pharma: Research Funding; Rhizen: Consultancy, Research Funding. Fisher:Dana-Farber Cancer Institute: Current Employment; Kyowa Kirin: Membership on an entity's Board of Directors or advisory committees. Haun:Kyowa Kirin: Membership on an entity's Board of Directors or advisory committees; Karger, Inc.: Patents & Royalties: Textbook royalties. Vermeer:Kyowa Kirin: Consultancy, Research Funding; Takeda: Research Funding; PIQUR: Research Funding; Innate Pharma: Consultancy. Ito:Kyowa Kirin Pharmaceutical Development, Inc.: Current Employment. Dwyer:Kyowa Kirin Pharmaceutical Development, Inc.: Current Employment. Herr:Kyowa Kirin, Inc.: Current Employment. Kim:Elorac: Research Funding; Eisai: Membership on an entity's Board of Directors or advisory committees, Research Funding; Corvus: Research Funding; Medivir: Membership on an entity's Board of Directors or advisory committees; Kyowa-Kirin Pharma: Research Funding; Innate Pharma: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Forty Seven Inc: Research Funding; Galderma: Membership on an entity's Board of Directors or advisory committees, Research Funding; Horizon Pharma: Consultancy, Research Funding; miRagen: Research Funding; Merck: Research Funding; Solingenix: Research Funding; Trillium: Research Funding; Takeda: Membership on an entity's Board of Directors or advisory committees, Research Funding; Neumedicine: Consultancy, Research Funding; Portola: Research Funding; Seattle Genetics: Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal